7th January, 2021 – The CARBALIVE Consortium (‘CARBALIVE’ or the ‘Consortium’) is pleased to announce positive data from the Phase 2 equivalent randomised, double-blind clinical trial of Carbalive™, a product candidate for the treatment of patients with decompensated cirrhosis. Carbalive™ is specifically designed to remove harmful bacterial toxins from the gut, reduce gut inflammation and its permeability, preventing them from leaking into the rest of the body.
The CARBALIVE consortium is focused on developing a novel treatment for patients with advanced liver disease. It is composed of partners from across Europe, led by University College London, and includes Yaqrit, a life sciences company and the intellectual property owner of Carbalive™ and the European Foundation for the Study of Chronic Liver Failure (www.efclif.com) among others. This academic-industrial consortium is funded by the European Union’s Horizon 2020 research and innovation programme.
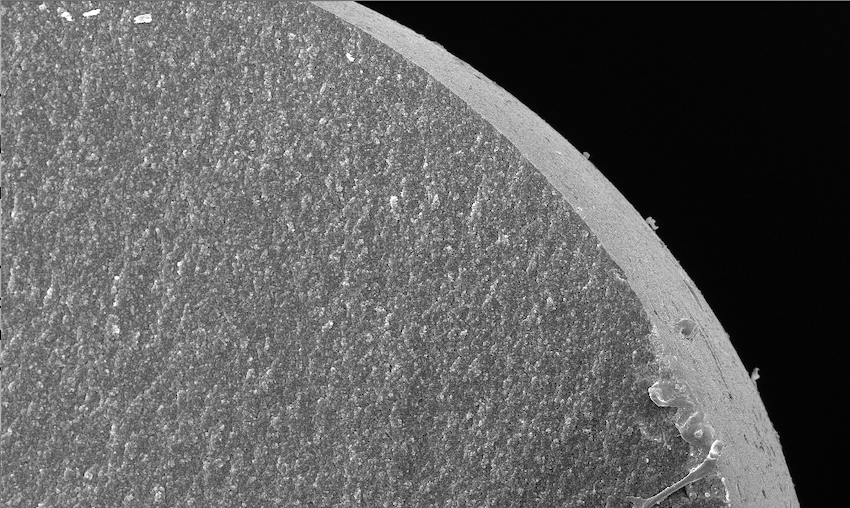
The Consortium is developing and testing Carbalive™, an orally-administered, non-absorbable engineered carbon of controlled porosity. The microscopic design of the Carbalive™ beads emerged from research into the importance of gut bacteria in the inflammation that accompanies cirrhosis. The research into the effects of the leakage of bacteria and bacterial products from the gut to the rest of the body, and how to inhibit it, was led by Professor Rajiv Jalan, Dr Jane Macnaughtan and members of the Liver Failure Group at University College London (UCL), and Yaqrit was spun off from UCL in 2014 to develop and potentially commercialise treatments including Carbalive™ for patients with advanced liver disease.
Carbalive™ has a special physical structure that adsorbs both large and small molecules in the gut. It is intended to treat patients with cirrhosis, a condition with over 100 million cases globally1 in 2017, accounting for the mortality of about 1 million patients each year.2 Patients with stable (compensated) cirrhosis from any cause, including alcohol, viral infection and fatty liver, can decompensate without warning, an event that usually leads to hospitalisation, is characterised pathophysiologically by systemic inflammation and carries a high risk of short-term mortality.
The randomised controlled, double-blinded clinical trial investigated the safety and tolerability of Carbalive™, compared to placebo, over a three month treatment period in adults with stable decompensated alcohol-related cirrhosis who had been abstinent from alcohol for the previous four weeks.
Carbalive™ was found to be safe, based on detailed clinical and biochemical assessments compared with the placebo and, was well tolerated with compliance of greater than 90%. Preliminary data from the trial also showed trends in the improvements of a wide range of biomarkers of systemic inflammation, which is especially notable because Carbalive™ is not systemically absorbed. Measures of gut-specific health also improved in Carbalive™ patients compared to those on the placebo arm. These improvements were associated with trends towards reduction in the markers of gut inflammation, less translocation (leakiness) of the gut wall and improvement of gut bacterial flora; characteristic problems associated with cirrhosis and its complications. The conclusions set forth in this announcement remain subject to finalising the remaining data analysis, the statistical and clinical study report and, therefore may change.
Professor Rajiv Jalan M.D., Professor of Hepatology at UCL, Founding Member of EASL CLIF and Project Coordinator for the CARBALIVE consortium, said, “With no specifically approved therapies for decompensated cirrhosis, there is a huge need for new treatment options. Results from this trial are very promising. The product’s safety and tolerability profile and, preliminary efficacy data continue to give us confidence that Carbalive is a promising candidate for the treatment of liver disease. Yaqrit is now planning to proceed with a pivotal trial in Europe, which could pave the way for regulatory approval in this region. I acknowledge gratefully the support of the EU H2020 grant, the huge contribution of all my collaborators and the patients who have been instrumental in the progress the Consortium has made in allowing the development of this treatment, that has the potential to save the lives of patients with cirrhosis.”
Professor Agustin Albillos M.D., of the Hospital Universitario Ramón y Cajal, Universidad de Alcalá, leader of the Liver and Digestive Diseases Group at the Ramón y Cajal Institute for Health Research-IRYCIS (Area 3) in Spain, said: “At its heart, this world leading consortium of experienced and expert research groups within the field of liver disease has sought to find a way to address the relationship between the gut’s microbiome and the progression of liver injury to advanced stages and complications of cirrhosis. These results reported today indicate that Carbalive is having a positive impact on inflammation and therefore could prevent this progression of cirrhosis complications. On behalf of the Consortium, I’d like to extend our gratitude to the EU’s Horizon 2020 programme, which has enabled us to bring this exciting new therapy to liver disease patients, who have few treatment alternatives.”
Yaqrit, the Carbalive™ intellectual property and commercial rights holder intends to conduct a randomised placebo-controlled European pivotal trial beginning in 2021. The Company plans to pursue investigation of Carbalive™ in further indications and to engage consultants in 2021 to advise as to the potential regulatory pathway in the US.
Full results from the trial will also be submitted for peer reviewed publication in a leading scientific journal.
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 634579 .
**ENDS**
For further enquiries:
For media enquiries:
Consilium Strategic Communications
Amber Fennell
Tel: +44 (0) 20 3709 5700
Email: fennell@consilum-comms.com
About Carbalive™
Carbalive™ is designed to remove harmful bacterial toxins from the gut before they leak to the liver. These engineered macroporous carbon beads, which are orally administered and designed to adsorb and remove lipopolysaccharides (LPS) from the gut without killing beneficial bacteria, are intended to prevent further progression and to treat decompensated cirrhosis (also known as decompensated liver disease), a condition with 10.6 million cases globally in 2017.
If an imbalance of bacterial toxins occurs in the gut, the gut may become inflamed and the gut wall may be compromised, causing the toxins to leak into the liver. In the liver, the toxins trigger a cascade of responses from the immune system that include inflammation and the death of liver cells. Over the long term, this may result in the scarring that is characteristic of cirrhosis and could ultimately progress to decompensation and liver failure. By removing the harmful toxins from the gut before they leak into the liver, Carbalive is intended to prevent recurrence or progression of liver decompensation in targeted patients with cirrhosis.
About the Carbalive Consortium
Humans have nearly 10 trillion intestinal bacteria. This is the gut microbiome, which both plays an important role in human health and is the largest reservoir of antigens and bacterial products in the body. If the gut is dysfunctional, as is the case in liver disease, the bacterial products can leak from the gut to the liver where they cause inflammation and injury. These changes happen in chronic liver disease (such as NASH) and in cirrhosis. In advanced cirrhosis, this bacterial translocation is linked to infections such as spontaneous bacterial peritonitis, which are associated with early death. Gut microbes also play a role in the progression of fatty liver disease to advanced liver disease, and it is these challenges that the Carbalive consortium was established to address. The Carbalive consortium has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 634579.
For more information visit www.carbalive.eu
About Yaqrit
Yaqrit is a clinical stage life sciences company focused on developing innovative treatments for patients with advanced liver disease in areas of high unmet medical need. The Company was founded by internationally renowned hepatologist Professor Rajiv Jalan of UCL, who has three decades of clinical, academic and research experience in treating patients with chronic liver disease. With his guidance, the Company has developed a clinical stage pipeline led by three programs: Carbalive and Dialive, both of which have complete randomised controlled clinical trials, and TLR4 antagonists, the most advanced of which is being developed by Akaza Bioscience, a joint venture with Takeda Pharmaceutical and Zenios Bioscience. These programs are designed to address unmet medical needs across the advanced stages of chronic liver disease from medically stable patients with decompensated cirrhosis in the outpatient setting, to critically ill, hospitalised patients with acute-on-chronic liver failure (“ACLF”). The application of the different therapeutic approaches to specific patient groups is based upon the Company’s deep understanding of the pathway and stratification of patients with chronic liver disease. For more information visit www.yaqrit.com
The contents of this announcement include statements that are, or may be deemed to be, “forward-looking statements”. In some cases, forward-looking statements can be identified by the use of forward-looking terminology, including the words “believes”, “estimates,” “anticipates”, “expects”, “intends”, “may”, “will”, “plans”, “continue”, “ongoing”, “potential”, “predict”, “project”, “target”, “seek” or “should” or, in each case, their negative or other variations or comparable terminology or by discussions of strategies, plans, objectives, targets, goals, future events or intentions. Forward-looking statements include statements regarding the consortium’s intentions, beliefs or current expectations concerning, among other things, prospects, growth, strategies and the industry in which the consortium operates. By their nature, forward-looking statements involve risks and uncertainties. Forward-looking statements are not guarantees of future performance. Given these risks and uncertainties, the reader should not rely on forward-looking statements as a prediction of actual results. Without prejudice to the consortium’s obligations under applicable law in relation to disclosure and ongoing information, the consortium does not intend, and does not assume any obligation, to update forward-looking statements.
References
Sepanlou S. et al. (2020). The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. The Lancet Gastroenterology & Hepatology.
Asrani et.al. Burden of Liver Disease in the World. J Hepatol 2019 Jan;70(1):151-171).

![]()
With no specifically approved therapies for decompensated cirrhosis, there is a huge need for new treatment options. Results from this trial are very promising.